How Endometriosis Systemic Disease Research Changes Your Treatment
Key Takeaways
1. Endometriosis is now understood as a whole-body, immune-driven inflammatory condition — not just a “women’s reproductive problem.”
2. Diet, exercise and mind-body practices are evidence-informed tools that can meaningfully reduce symptoms — especially when combined with medical care.
3. The treatment pipeline is evolving fast, with new oral medications, targeted implants and microbiome research offering real hope for more personalised, less side-effect-heavy options in the near future.
Introduction
If you’ve been told your pain is “just a bad period,” you deserve better. The latest endometriosis systemic disease research from 2023–2026 has fundamentally changed how we understand this condition. No longer viewed as just a gynecological issue, scientists now recognize it as a complex, whole-body inflammatory process affecting one in nine Australians.
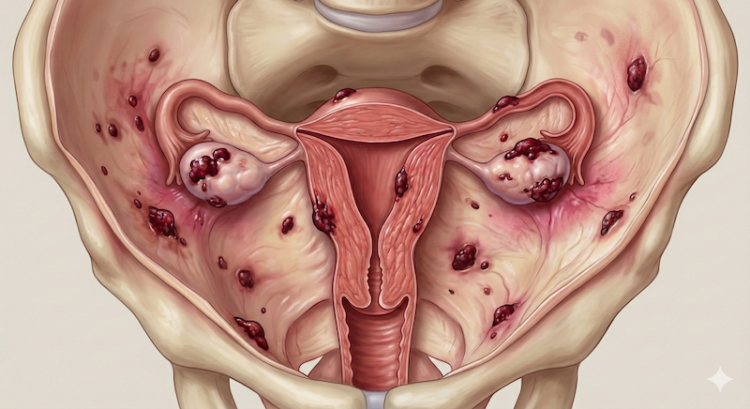
What Endometriosis Actually Is — and Why the Old Story Was Incomplete
For decades, endometriosis was described almost exclusively as a gynaecological condition: tissue similar to the lining of the uterus growing in places it shouldn’t — typically on the ovaries, fallopian tubes, and pelvic lining. It was treated as a localised problem with localised solutions: surgery to remove lesions, hormonal therapies to suppress the cycle, and if all else failed, a recommendation to manage and endure.
That framing is now understood to be incomplete, and for many people living with the condition, that understanding comes as a profound — if belated — validation.
What the most current research tells us is that endometriosis is a systemic, whole-body disease. It involves complex, ongoing interactions between your hormonal system, your immune system, your nervous system, and increasingly, your gut microbiome. The World Health Organization’s 2025 fact sheet explicitly identifies immune dysregulation and chronic inflammation as key features of the disease — not just side effects, but central drivers.
This matters for you because it reframes what’s happening when you experience widespread pain, fatigue, bowel disruption, brain fog or mood changes. These aren’t separate problems sitting alongside your endometriosis. In many cases, they are endometriosis — expressing itself through the body-wide inflammatory processes that characterise the condition at its core.
Understanding this also changes what a meaningful treatment approach looks like. It’s no longer sufficient to think only about the pelvis. Managing endometriosis well increasingly means thinking about your immune health, your gut, your nervous system, your diet, your stress load, and your overall inflammatory baseline.
The Immune System: Your Body Turned Against Itself
At the heart of the new endometriosis science is a clearer picture of how your immune system gets involved — and how it ultimately fails to do its job.
In a healthy body, when endometrial-like cells travel outside the uterus, the immune system identifies and clears them. In endometriosis, this clearance mechanism appears to break down. Macrophages — the immune cells responsible for cleaning up unwanted tissue and debris — become dysregulated. Instead of eliminating the misplaced cells, they shift into a mode that promotes inflammation, encourages the growth of blood vessels that feed lesions, and effectively helps the disease establish and sustain itself.
This isn’t a failure of willpower or lifestyle. It’s a failure of immune regulation at a cellular level.
Research from 2024 and 2025 has also implicated bacterial signals — specifically a molecule called lipopolysaccharide (LPS), found on the surface of certain gram-negative bacteria — in activating the immune pathways that drive endometriosis progression. This means what’s happening in your gut and reproductive tract microbiome may be directly feeding the inflammatory fire in your pelvis.
For you as someone navigating this condition, this immune-centred model has one important immediate implication: anything that reduces your overall inflammatory load may help. Not cure — but help. Diet, sleep, stress, and movement all influence immune function. They are not alternative medicine. They are biology.
Your Gut Has More to Do With This Than You Might Think
One of the more surprising frontiers in endometriosis research over the past two years has been the microbiome — the vast community of bacteria, fungi and other microorganisms living in your digestive tract and reproductive system.
Multiple studies and a 2025 scoping review have now identified consistent patterns of microbial imbalance in women with endometriosis. Compared to those without the condition, you’re more likely to have fewer bacteria that produce short-chain fatty acids (SCFAs) — particularly a compound called n-butyrate — and more bacteria associated with inflammation and tissue invasion, including Fusobacterium nucleatum, which has been shown in mouse studies to actively promote lesion growth and fibrosis.
N-butyrate matters because it has a direct, measurable suppressive effect on endometriotic cells. In laboratory studies, higher butyrate levels inhibit the proliferation of these cells. Lower butyrate levels — which appear more common in people with endometriosis — may allow unchecked growth.
No single validated “endometriosis microbiome signature” has been established yet, and this area of research is still young. But the pattern is consistent enough that researchers are now actively exploring microbiome-targeted therapies: restoring SCFA-producing bacteria, modulating pro-inflammatory species, and investigating whether diet-driven shifts in gut flora might alter disease progression.
What this means for you right now is nuanced. It doesn’t mean that taking a probiotic will treat your endometriosis. It does mean that the connection between your gut health and your pelvic health is real, biologically grounded, and worth taking seriously as part of your overall management strategy — in conversation with your healthcare team.
The Australian Landscape: More Investment, But Barriers Remain
If you’re managing endometriosis in Australia, it’s worth knowing that the research and policy environment has shifted meaningfully in recent years — and that this is partly the result of sustained advocacy from people with lived experience of the condition.
Australia’s 2024 Endometriosis Progress Report documents more than $19 million in Medical Research Future Fund grants directed across eleven endometriosis projects. These cover a broad range, from earlier diagnostic techniques and pain mechanism research to imaging innovation and cell biology. The NECST Network — the National Endometriosis Clinical and Scientific Trials network — and the EndoZone patient information hub have both received continued funding, representing a more coordinated national approach than existed even five years ago.
Specialist centres like the Julia Argyrou Endometriosis Centre at Epworth Hospital are running active clinical trials into new medications, surgical approaches and models of care. A 2025 Monash University study tracking almost 20,000 women aged 14 to 49 reported a significant rise in recorded endometriosis diagnoses over time — interpreted not as an explosion in actual incidence, but as better recognition and coding by clinicians. People are being diagnosed. That is progress.
And yet. Diagnostic delay — the gap between when symptoms first appear and when you finally receive a confirmed diagnosis — remains one of the most painful aspects of living with this condition. Access to specialist care is uneven, with significant disparities between metropolitan and regional Australia. The research investment is welcome and real, but the lived-experience gap has not closed.
Knowing this context matters because it helps you navigate the system with realistic expectations — and understand that the delays and dismissals you may have encountered reflect systemic failures, not your own.
The Evolving Treatment Landscape: What’s New and What’s Coming
Hormonal Therapies — Better Options Are Emerging
Surgery and hormonal suppression remain the backbone of endometriosis treatment, but the hormonal side of that equation is becoming more sophisticated. A 2025 analysis of global clinical trials shows strong momentum behind a class of drugs called oral GnRH antagonists — including elagolix, relugolix and linzagolix — which work by reducing estrogen production in a more controllable, reversible way than older injectable options.
Phase 3 trial data — including the EDELWEISS 3 trial for linzagolix — show clinically meaningful reductions in both period pain and non-menstrual pelvic pain when these drugs are combined with low-dose “add-back” hormones, which help offset some of the menopausal-type side effects. Bone mineral density loss and symptoms like hot flushes remain concerns that require ongoing monitoring, but the ability to fine-tune the dose orally represents a genuine improvement in tolerability and quality of life for many people.
The era of one-size-fits-all hormonal suppression is, slowly, giving way to something more personalised.
Local Delivery: Treating the Problem Where It Lives
One of the more promising developments from 2024 is a Yale pilot project testing long-acting biodegradable implants designed to release estrogen-blocking medication directly at the site of endometriotic lesions. The goal is to achieve the therapeutic effect of hormonal suppression while dramatically reducing the systemic side effects that make current treatments so difficult for many people to sustain.
This approach is still early-stage, but it reflects a broader shift in thinking: rather than flooding the entire body with hormone-altering drugs, can we deliver treatment with precision? If the research continues to progress, local-delivery systems could eventually offer a meaningful alternative for people who struggle with the systemic burden of current hormonal therapies.
Non-Hormonal Frontiers: Immune and Microbiome Targets
Beyond hormones, early translational research is exploring non-hormonal strategies that target the immune and microbial mechanisms described earlier in this article. These include approaches aimed at restoring SCFA levels, modulating pro-inflammatory bacteria, and directly interfering with the TLR4–LPS signalling pathways that appear to activate macrophage dysfunction.
None of these is close to clinical use yet. But they represent a fundamentally different approach to the disease — one that doesn’t rely on hormonal suppression at all, and that could eventually offer options for people who cannot tolerate or do not respond to existing therapies.
What You Can Do Now: Lifestyle Medicine and the Evidence Behind It
This is where many people with endometriosis have the most immediate agency — and where the research has moved most visibly in the past two to three years. The evidence is not perfect. Many studies are small, short-term or heterogeneous. But the direction of the findings is consistent, and the biological rationale is strong.
Eating in a Way That Supports Your Body
A 2025 case-control study found that women with higher adherence to a Mediterranean-style diet had dramatically lower odds of endometriosis — an adjusted odds ratio of 0.06, meaning those eating a Mediterranean-style diet were associated with dramatically lower rates of the condition compared to those who weren’t. Higher meat and dairy consumption, and lower intake of fruits, vegetables, legumes and fish, was associated with greater risk.
What does this look like in practice? A Mediterranean-style, anti-inflammatory diet centres on:
- Abundant vegetables, fruits, legumes and whole grains
- Regular fish — particularly oily fish rich in omega-3 fatty acids
- Olive oil as your primary fat source
- Nuts and seeds
- Minimal red and processed meat
- Low intake of ultra-processed foods and refined sugars
This isn’t a cure. It’s an evidence-informed approach to reducing your overall inflammatory burden through food — and the research increasingly supports it as a reasonable, beneficial component of comprehensive endometriosis care.
Specific supplements also show promise in smaller trials, including omega-3 fatty acids, vitamins C, D and E, magnesium, curcumin (the active compound in turmeric), zinc, folate, N-acetylcysteine, and green tea polyphenols. The evidence base for each is still developing, and dosing guidelines are not yet robust — but discussing these with a healthcare professional or dietitian is a reasonable step.
Managing Gut Symptoms With a Low FODMAP Approach
If bowel symptoms are a significant part of your experience — bloating, abdominal pain, constipation, altered stool — a low FODMAP diet may offer meaningful relief, and the evidence for this has strengthened considerably.
A 2025 randomised controlled crossover trial of 35 women with endometriosis and significant gastrointestinal symptoms tested a 28-day low FODMAP diet against a matched control diet. 60% responded on the low FODMAP diet, compared to 26% on the control — a statistically significant difference. Global GI symptoms, abdominal pain, bloating, stool consistency and both gut-specific and endometriosis-specific quality of life all improved meaningfully.
A complementary 2025 real-world study found that among endometriosis patients who completed a low FODMAP program, constipation and several quality-of-life domains improved, and approximately two-thirds reported less pain — particularly chronic pelvic pain.
This is not a diet to adopt indefinitely or without support. Low FODMAP is designed to be time-limited — typically four to eight weeks for the elimination phase — and should be undertaken with the guidance of a dietitian who can supervise the reintroduction phase and ensure you’re not compromising your nutritional intake in the long run.
Moving Your Body: What the Research Shows
Physical activity influences endometriosis through several overlapping pathways: it lowers systemic inflammatory markers, modulates estrogen metabolism, supports immune regulation, and has well-documented benefits for pain perception, mood and psychological resilience.
Small interventional studies have shown improvements in pain, dyspareunia (pain with intercourse) and quality of life from regular exercise, though not all have reached conventional statistical significance. A supervised nine-week program combining stretching and strengthening produced significant gains in quality of life and meaningful reductions in dyspareunia among its participants.
You don’t need to train like an athlete. Gentle, consistent movement — walking, swimming, yoga, Pilates — appears to be sufficient to capture meaningful benefit. If pain is currently limiting your capacity to exercise, that’s worth raising with your doctor or a physiotherapist who specialises in pelvic health.
Mind-Body Strategies: Taking Your Nervous System Seriously
Chronic pain is not just a physical event. It involves central sensitisation — the nervous system becoming progressively more attuned and reactive to pain signals over time. For people with endometriosis, this means that managing the nervous system is as legitimate a treatment target as managing the lesions themselves.
An overview of lifestyle interventions in endometriosis found that yoga and broader mind-body programs have some of the more robust — though still small-scale — evidence for reducing pain and improving coping. Mindfulness-based stress reduction, somatic therapies and targeted psychological support for chronic pain have all shown benefit across pain conditions generally, with emerging application to endometriosis specifically.
This isn’t about being told your pain is “in your head.” It’s about recognising that your nervous system is a modifiable part of this disease, and that giving it the right inputs — rest, breath, movement, connection, meaning — is a clinical strategy, not a luxury.
Putting It All Together: A Whole-Person Framework
What the 2023–2026 research wave is building toward is a genuinely integrated model of endometriosis care. One that holds the complexity of this condition — its hormonal drivers, its immune character, its microbial connections, its neurological dimensions — and responds to that complexity with equally multi-layered care.
For you, this means that an honest conversation about your endometriosis management might include your gynaecologist, a dietitian, a pelvic floor physiotherapist, a pain psychologist, and your GP — each addressing a different system that the disease affects. It means that your diet is not a soft, optional lifestyle choice but a legitimate tool with biological mechanisms behind it. It means that your gut symptoms deserve investigation, not dismissal. And it means that emerging treatments — from oral GnRH antagonists to local-delivery implants to microbiome-targeted therapies — offer a real and expanding horizon of options.
You are not at the mercy of this condition. The science is catching up, the investment is increasing, and the framework for thinking about endometriosis is becoming more sophisticated and more human at the same time.
Frequently Asked Questions
Q1: Can changing my diet actually make a difference to my endometriosis symptoms?
Yes — with an important caveat about what “make a difference” means. Diet cannot remove lesions or reverse the underlying disease, but it can meaningfully influence your body’s inflammatory environment, gut microbiome composition, hormonal metabolism and pain experience. The 2025 Mediterranean diet case-control study and the low FODMAP RCT both provide solid support for dietary intervention as a symptom management tool. The strongest evidence points toward a Mediterranean-style, plant-forward eating pattern as a general baseline, with a supervised low FODMAP approach for those with significant bowel symptoms. Working with a dietitian who understands endometriosis will help you tailor this to your specific situation rather than applying a one-size-fits-all protocol.
Q2: What are GnRH antagonists and how are they different from the hormonal treatments I might already be on?
GnRH antagonists are a class of medications that work by blocking the brain’s signals to produce the hormones that drive estrogen production. Unlike older GnRH agonists (such as Zoladex), which cause an initial hormone flare before suppressing production, antagonists act immediately and allow more precise dose adjustment. The newer oral options — elagolix, relugolix and linzagolix — offer the added convenience of a daily pill rather than an injection, and can be combined with low-dose “add-back” hormone therapy to manage side effects like bone density loss and menopausal-type symptoms. If you’ve previously stopped a hormonal treatment due to side effects, it may be worth asking your specialist whether one of the newer oral GnRH antagonists might be a better fit.
Q3: Is there a link between my gut symptoms and my endometriosis, or are they two separate problems?
The evidence increasingly suggests they are connected — and not just coincidentally. People with endometriosis show consistent patterns of gut microbiome dysbiosis, including reduced levels of bacteria that produce anti-inflammatory short-chain fatty acids and increased levels of pro-inflammatory species. Direct lesion involvement of the bowel is also common in certain forms of endometriosis. This means your gut symptoms deserve thorough investigation — including a conversation with a gastroenterologist or a dietitian experienced in both endometriosis and digestive health — rather than being treated as a separate, unrelated issue. Addressing gut health may not only relieve bowel symptoms but potentially influence the broader inflammatory environment driving your endometriosis.
Q4: I’ve read that stress makes endometriosis worse. Is that actually true, or is it just another way of blaming lifestyle?
The connection between stress and endometriosis is biologically real, not a moral judgement. Chronic psychological stress activates the hypothalamic-pituitary-adrenal (HPA) axis and the sympathetic nervous system, driving cortisol dysregulation and systemic inflammation — both of which are relevant to endometriosis pathology. Additionally, endometriosis involves central sensitisation of the nervous system, meaning chronic pain itself reshapes how your nervous system processes future pain signals. Stress-reduction practices — yoga, mindfulness, somatic therapies, therapeutic support — are therefore not soft options or lifestyle extras. They are interventions targeting a biological system that is genuinely involved in your disease. That said, stress management is an adjunct to, not a replacement for, medical care — and the responsibility for managing this disease does not rest on your ability to stay calm.
This article is intended for informational purposes and does not constitute medical advice. If you are experiencing symptoms of endometriosis or are currently managing a diagnosis, please speak with a qualified healthcare professional about the options most appropriate for your individual circumstances.